Gallium Solar Panel
A solar panel installer requested the following quote:
UniversityWafer, Inc. Quoted:
We have 17 containers 460 Watt solar panels ready to pick up in Long Beach warehouse now.
Certification: UL/CEC/FSEC. Warranty: 25 Years.
Reference #271303 for specs and pricing.
What is the Efficiency of GaAs Solar Cells
Buy GaAs Wafers Online or Send Us Your Specs!
In the case of single-junction solar cells, the Gallium Arsenide GaAs solar cell showed an efficiency of 24.3% - the highest value ever reported for a single-junction solar cell. This efficiency record (24 - 3%) was achieved by deposition of the III - V semiconductor layer directly on silicon, and the efficiency of the wafer bond production silicon on gas silicon (S-S) cell increased to 34.1%. The efficiency rate of cultured GaAs solar cells on silicon wafers was 29-1%, but the efficiency for both silicon and silicon oxide cells (SiO2) was equally high at 29.2% and 28.5%, respectively. We have achieved a new record in the efficiency of the individual transitions of a silicon / GaAs cell, "said lead author Dr J.J. Pappalardo from the Department of Materials Science and Engineering at Imperial College London. [Sources: 1, 2, 4, 5]
But even this fact makes it possible for gallium arsenide solar cells to produce only a fraction of the efficiency of their silicon counterparts. As a GaInP alternative, the average efficiency rate of a solar cell grown on HPV is estimated at 27%. [Sources: 5, 10]
InGaP - triple-based solar cells that are widely used in space and have a record efficiency of 28.8%, but still have a limited efficiency of 30-32% (as shown in Figure 2). Multiple solar cells have been developed with a potential efficiency of between 30% and 32%. This is significantly lower than gallium arsenide, whose single compound solar cells have a potential efficiency of less than 32% (Figure 3). GaInP-based 3-junction solar cells - which have record efficiencies of up to 28-8% - are widely used in space. Multi-junction solar telephones are being developed, although their limit value is between 30 and 32%. [Sources: 4, 7]
These multi-junction solar cells are intended to power electric vehicles and contribute to the solar cell for room concentrators. III - V composite solar cells are used in space because they have a higher efficiency and radiation resistance than Si solar cells (as illustrated in Figure 19-44). They are more efficient than simple composite cells and can be used for solar telephones, but have a lower energy density and higher thermal conductivity than SiSolar cells. II - III Composite solar phones, but they are likely to make a contribution and are not expected for electric vehicles - solar panels with electricity [Sources: 4]
A highly efficient solar cell made of gallium arsenide would potentially take electric vehicles off the grid and allow electronics to generate energy from ambient light, including indoor lighting. [Sources: 7]
In fact, it is not only gallium that bodes well, but arsenide, which is the range of gallium arsenids. There is no other material in the world with such a high bandwidth for solar cells, and it is actually the only one capable of this type of highly efficient solar cell. [Sources: 0, 3]
The maximum theoretical efficiency that a single bandgap solar cell can achieve with unconcentrated sunlight is around 33.5%. Compared to silicon solar cells, GaAs solar cells have an efficiency advantage of about 40% compared to their silicon counterparts [1]. Taking efficiency benefits into account, the total energy collected from a hot gallium rooftop solar array can be up to 50% higher than that of competing silicon solar panels. [Sources: 7, 8, 9]
There are currently only a few types of solar cells that can produce as much energy per unit of mass as gallium arsenide (GaAs). The development of new technologies such as hydrogenated silicon thin-film solar cells could reduce production costs per unit of mass (per power) by orders of magnitude compared to current levels. [Sources: 6, 10, 11]
With one of the means discussed, gallium solar cells can demonstrably generate more energy per unit of mass (per power) than any other type of solar cell. With a discussion, they can prove the existence of a new class of high-cost, efficient photovoltaic cells (PV) with a high capacity of up to 1,000 watts per square meter (kW / m2) and low costs of around $10 per kilowatt hour (KWh) per year. With a means under discussion, less than a third of the energy - per unit - can be generated by - mass of other types of solar cells. [Sources: 10]
We chose the solar cells we discussed because we can rely on the sun to continue shining and to provide solar power. We chose them because they can rely on mass, not only the solar cell itself, but also a number of other types of photovoltaic cells such as solar panels and solar thermal systems. We chose them because they can rely on their mass, which we can rely on not only to maintain their shine and generate solar power, but also on any other type of PV cell. [Sources: 10]
The main task of this study is to determine whether silicon-gallium-arsenide solar cells are suitable for missions with solar probes and to determine the feasibility of using such solar cells for missions with solar probes. In this article we will outline how to create one, as well as possible development paths that will continue in the future. This chapter describes the development of GaAs - based on single-connection solar cells - and the process of realizing highly efficient solar telephones and solar modules. [Sources: 4, 6, 10]
Sources:
[0]: https://www.frontiersin.org/articles/10.3389/fchem.2019.00137/full
[1]: https://www.photonicsviews.com/new-records-for-triple-junction-solar-cells/
[2]: https://optics.org/news/9/12/19
[3]: https://www.universitywafer.com/gaas-solar-cell-pros-cons.html
[4]: https://www.intechopen.com/books/post-transition-metals/high-efficiency-gaas-based-solar-cells
[5]: https://pv-magazine-usa.com/2020/01/13/solar-cells-from-space-are-on-the-way/
[6]: http://escapulario.com.br/js/3ibe20/452997-gallium-arsenide-solar-panels
[7]: https://medium.com/rayton-solar/5-ways-gallium-arsenide-solar-cells-outperform-silicon-78b479c0bc
[8]: https://www.techbriefs.com
[9]: https://www.hindawi.com/journals/ijp/2017/8269358/
[10]: https://www.appropedia.org/Gallium_arsenide_solar_cells
[11]: https://www.encyclopedia.com/science/news-wires-white-papers-and-books/solar-power-systems
Why is Gallium Arsenide Better Than Silicon in Solar Cells?
Gallium arsenide (GaAs) is a semiconductor material that is often used in the production of solar cells. GaAs  solar cells are known for their high efficiency, which means that they can convert a higher percentage of sunlight into electricity than other types of solar cells. They are also able to operate at higher temperatures than traditional silicon solar cells, which can help to improve their performance. In addition, GaAs solar cells are relatively lightweight and have a high tolerance to radiation, making them suitable for use in a variety of applications, including space-based solar power systems.
solar cells are known for their high efficiency, which means that they can convert a higher percentage of sunlight into electricity than other types of solar cells. They are also able to operate at higher temperatures than traditional silicon solar cells, which can help to improve their performance. In addition, GaAs solar cells are relatively lightweight and have a high tolerance to radiation, making them suitable for use in a variety of applications, including space-based solar power systems.
In the last few years, advances in gallium arsenide solar cells have been making headlines. The first Mars rovers, Opportunity and Valor, used these devices to collect energy during their visits. These rovers were the first human missions to use solar cells to power their equipment. But after 20 years of operation, the conversion rate of the solar cells dropped dramatically, primarily due to dust storms on the surface of Mars.
The American Opportunity rover uses nearly 500 gallium arsenide solar cells, which have a relatively low 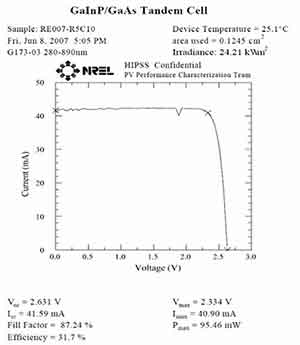 conversion rate (less than 30%) - not ideal in space, but more cost-effective than any other power supply. The United States has also used solar cells on its InSight Mars lander. While nuclear batteries are a better option for the long term, they are prohibitively expensive and require a huge one-time investment.
conversion rate (less than 30%) - not ideal in space, but more cost-effective than any other power supply. The United States has also used solar cells on its InSight Mars lander. While nuclear batteries are a better option for the long term, they are prohibitively expensive and require a huge one-time investment.
The gallium arsenide solar cell has several advantages over its silicon counterpart. It is more durable and resistant to moisture, radiation, and ultraviolet light. Therefore, it is better for use in space, where the temperatures can be extremely high. A single sliver of gallium arsenide solar cell can be used to charge an electronic device while remaining lightweight and flexible. This sliver of gallium is much cheaper than silicon.
The best solar cells will use a combination of crystalline and noncrystalline silicon materials. A gallium arsenide wafer is more efficient than silicon because it can absorb more sunlight than a silicon wafer. It can also be used on a thin flexible substrate, ensuring it is portable and lightweight. The high efficiency level of gallium arsenide solar cells make it the best choice for charging electronic devices without grid electricity.
A single-junction gallium arsenide solar cell is more efficient than silicon solar cells. This is why NASA uses GaAs for its high-frequency radio signals. Its high efficiency is another reason why the technology is so popular. The best GaAs cell will allow a greater amount of solar energy to be converted into electrical energy. This is an advantage of solar cells. A crystalline gallium-arsenide wafer can produce more energy than a silicon-based panel.
The performance of gallium arsenide solar cells depends on their composition. The best gallium arsenide solar cells have an output power of 460 watts at room temperature, which is lower than the output power of silicon crystals. However, this is not the optimal power supply mode for Mars. It is an ideal power source for the moon. The United States also used them on its InSight Mars lander.
The performance of gallium arsenide solar cells depends on the material used for the solar cell. While most solar cells are produced using silicon crystals, gallium arsenide solar cells are more efficient. This means that gallium ados solar cells are more cost-effective. They will also generate more electricity than a lithium-ion cell, but are not as effective as gallium-arsenide panels.
The gallium arsenide solar cell performs better than its silicon cousins in various situations. For instance, it can operate under conditions of 250 degrees Celsius while silicon-based solar cells will stop working at about 200 degrees C. This is an excellent advantage for astronauts because they can use solar cells in harsh environments. This type of solar cell is also cheaper and easier to produce than silicon-based ones. It has a longer lifespan and can be installed anywhere, and it will also last longer.
The gallium arsenide solar cell is superior to silicon solar cells in a wide range of applications. It can withstand higher temperatures and can function in extreme environments. The American Opportunity rover, for example, has 4 big wings. The US InSight Mars lander has successfully used three-junction gallium ados solar cells. The Chang'e 3 and Chang'e 4 rover's slender design allows for larger battery capacity.
Aside from being cheap, GaAs solar cells have impressive performance. Despite their large size, they are expensive to manufacture and have been used mostly for satellites and spacecraft. But they aren't the only applications for GaAs solar cells. There are many other ways to use these solar cells, including installing them on buildings. They can provide energy in remote locations or on rooftops. Their high efficiency makes them a great alternative for solar power systems.
What are the Pros/Cons of Gallium Arsenide Solar Cells
 Despite its many advantages, gallium arsenide can be toxic to animals and humans, and thus it needs to be regulated for human consumption. In order to be used in solar cells gallium arsenide must first be filtered. The filtering process removes any particles that are larger than 10 micrometers. At this size particles bind to gallium atoms when the gallium ions are present, and prevent them from being reabsorbed into the bloodstream when gallium ions are passed through the intestinal tract.
Despite its many advantages, gallium arsenide can be toxic to animals and humans, and thus it needs to be regulated for human consumption. In order to be used in solar cells gallium arsenide must first be filtered. The filtering process removes any particles that are larger than 10 micrometers. At this size particles bind to gallium atoms when the gallium ions are present, and prevent them from being reabsorbed into the bloodstream when gallium ions are passed through the intestinal tract.
In order for gallium to be effectively filtered, it is mixed with a liquid solution that consists of boron. The combination of boron and gallium thioglycolate filters large particles that might be attached to gallium atoms when gallium ions pass through. This mixture also prevents the aggregation of gallium molecules into larger molecules that could be hazardous to humans. This filtering procedure is often combined with a step that breaks down the gallium into simpler compounds that can be easily filtered through membranes in solar cells.
One of the most common uses for gallium arsenide in solar cells is in the creation of black silicon, which is useful in semiconductor technology. It is also used to prevent the emission of electron beams from solar cells in solar photovoltaic cells. The reason for this is that black silicon has a positive charge and electrons are positively charged. Electrons are always attached to one side of a positively charged surface. Gallium can change the alignment of the electrons so that the surface can be more positively charged and thus trap more electrons than a surface that is evenly neutral.
Gallium arsenide can be combined with other materials in solar cells to create a material that will be highly conductive of electricity. Silicon is one material that has this feature. However, gallium together with other substances can also create materials that are highly attractive to the eyes of the human eye. These materials are called solar cells. Because the eyes are designed to be attracted to bright colors, solar cells can help reduce glare in the evening and help to prevent eyestrain.
Gallium arsenide can be combined with other materials to form thin films that can be used in making thin films of gallium arsenide that can be placed on the surface of other materials. Gallium combines extremely well with boron and other materials. When gallium combines with these compounds, it creates new materials that are even better conductors of electricity than gallium mixed with silver. The combination of gallium and boron creates new types of solar cells that can be used in electronic devices that use semiconductors like semiconductor diodes.
Gallium arsenide is so similar to silver that many people mistakenly think that it is made from the same element. Gallium was actually discovered when scientists tested different types of gases. They noticed that when they used gallium combined with certain gases they produced a type of gas that is very similar to silver.
Gallium arsenide is a useful metal for making solar cells because it is abundant in many locations around the world. Gallium is one of the most plentiful elements in the earth's crust and is found in such places as granite, which is often used in building construction. It also is found in many different types of rocks including limestone. It is also commonly found in coal, which is one of the world's most common sources of energy.
As you can see gallium arsenide is an interesting material to have as part of your solar cells. Gallium is a light-weight material and is good for making solar cells that can be used in a variety of devices. Gallium arsenide solar cells are fairly inexpensive as well. Because of these factors gallium arsenide, gallium, and gallium arsenide, solar cells can be purchased relatively cheap.
How Much Power Do Gallium Solar Panels Generate?
Gallium arsenide is a useful metal that can be used in solar cells. It is abundant throughout the world and is considered to be one of the best semiconductor materials. Because gallium arsenide solar panels are so expensive, it is not a practical choice for mass production. In the meantime, researchers are working to develop cheaper alternatives to gallium and silicon. However, the materials used in these panels are not perfect. There are flaws that should be considered before deciding on a panel's design.
Gallium arsenide is a highly efficient material for solar cells and is used in NASA's InSight Mars lander. It is  not as efficient as silicon crystals but is more attractive to human eyes. Although this type of solar cell is not ideal for the red planet, it is still an excellent choice for other places. The United States used these cells in its InSight lander on Mars to provide power to the lander.
not as efficient as silicon crystals but is more attractive to human eyes. Although this type of solar cell is not ideal for the red planet, it is still an excellent choice for other places. The United States used these cells in its InSight lander on Mars to provide power to the lander.
Another way to make solar panels use gallium is to combine the material with another substance. The combination of the two elements creates a highly efficient material with high energy yield. In fact, gallium arsenide can be more efficient than silicon. This makes it an excellent choice for solar energy because it is rarer than gold and it is also toxic. The cost of the materials is very low and the cost of solar panels is expected to fall further as more developments are made.
Gallium arsenide solar cells are much better than their silicon cousins. The best gallium arsenide solar cells can operate at a temperature of 250 degrees Celsius, while silicon cells will eventually stop working even under these conditions. This makes them ideal for the moon and Mars, as well as other harsh environments. Since the materials used in these panels are so expensive and rare, the use of these panels on space missions is a smart move.
Gallium arsenide solar cells are more efficient than silicon solar panels. They have a higher conversion rate than silicon, which is the most common material used in solar cells. Because they are more expensive, gallium arsenide can't be mass-produced and is too rare for mass use. In general, solar cells using this material are better than silicon-based panels, but they are more expensive.
The cost of gallium arsenide solar panels is higher than those made from silicon. Moreover, the material itself is expensive. But, these panels can be used in many applications, including building energy-efficient homes. The materials are more efficient than silicon-based solar panels, so they're a good option for remote locations and even for space exploration. If you are looking for a cheap solar panel, you can opt for the gallium arsenide ones.
This material is more durable than its silicon counterpart. It can withstand extreme temperatures and can be used in remote areas. It is also more durable than its silicon cousin. This is great news for the environment, and it's also cheaper to produce than its silicon cousin. And since it can be used anywhere, it's a great choice for home-use. It can be installed anywhere if you need to, even in rural areas.
Solar panels made of gallium arsenide contain a layer of gallium and arsenic that can produce a lot of energy. Because it is rarer than gold, it can be extremely expensive. Its high cost and high risk make it unsuitable for most people, but it is still the best choice for many. In the future, solar power will be more affordable than its current cost.
While gallium arsenide solar panels can be expensive, they are still among the most affordable forms of solar energy. They are made of silicon, which is the main material used in solar panels. Unlike gallium arsenide, it is cheap to produce and use in many other applications. If you're looking for a solar panel that doesn't cost a lot, the only thing that matters is that it will generate energy.
