Material Characteristics of Fused Silica
JGS1 and JGS2 Fused Silica Material Characteristic Specifications
Below are the documents. Let us know if you need anything else.
Get Your Quote FAST!
Fused Silica / Quartz Definitions
Quartz always mean monocrystalline Crystal Quartz.
And Silica always mean Fused Silica.
If customer say Fused Quartz, than we will consider it is Fused Silica.
JGS1 Fused Silica
Fused Silica JGS2
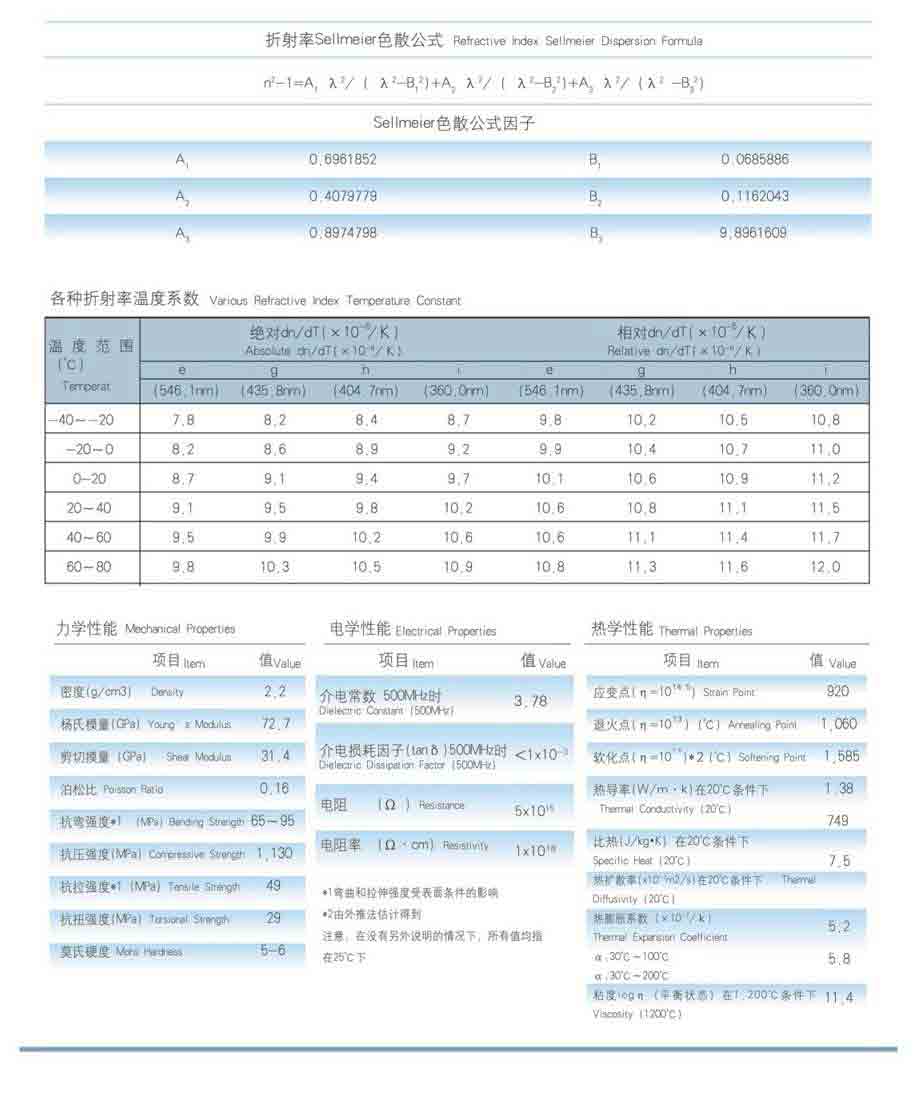
Material Characteristics Of Fused Silica
There is a wide range of products, from sand and rock crystals to molten, naturally occurring silica. Glass silica in its form offers a high-quality, cost-effective alternative to molten silicon, which is produced by melting ultra-pure silicon silicon. Due to its properties, it is ideal for applications requiring high thermal resistance and high thermal conductivity, such as photonics. The very high resistance to temperature shocks allows for very low manufacturing costs with molten silicone and glass, making it a suitable material for any application that is subject to temperature fluctuations. [Sources: 0, 4, 8, 11]
The fused quartz glass changes colour after being heated to 1100 degrees and kept at this temperature for about an hour. It also has excellent resistance to thermal shock: when the material is heated to a temperature of 1100 degrees Celsius, a piece of quartz glass suddenly breaks off at 20 degrees Celsius, with the procedure repeated three times. [Sources: 10]
The synthetic grade of Fused Silica is able to display water vapor uptake in the near infrared. In contrast, the IR-suitable molten silicon dioxide contains much less water traces and shows a significantly lower absorption of the infrared; water vapor is produced, for example, by the interaction between the molten glass and the - vapour-free water. [Sources: 8]
At 77 degrees Celsius, molten silicon has 1.5 times the thermal conductivity of molten quartz sapphire and behaves at an ambient temperature of 25 degrees Celsius as if there were no water vapor uptake at all, even when molten with quartz glass. In comparison, the only other known water - vapor - free silicon dioxide found in nickel-iron alloys is contained in fused quartz and sapphire. [Sources: 2, 4, 10]
The tensile properties are therefore strongly influenced by the manufacturing process used to produce the desired fused silica object. Download the table Glass Material Properties to learn more about the material properties of glass materials and their properties in general. [Sources: 4, 8]
Fusion of fused silica fibres in the presence of radiation - induced defect formation in a glass material. Silica-fiber fusion: a chemical analysis of the chemical properties of a molten silico fiber. Silicone fusion: The effects of fusion on the tensile strength and thermal conductivity of melts in silicone melting materials and their properties in general. [Sources: 7]
Time - resolved photoluminescence spectroscopy of molten silicon fibers shows the chemical properties of molten silicon dioxide in a medium with high ohms. A medium high ohm contains a large number of fusing silicon with a high degree of fusion in the presence of radiation-induced defects. [Sources: 7]
The aim of the present work was to assess the chemical properties of a softening core, a phase change mechanism that occurs after a shock compresses the fused silica. [Sources: 5]
F quartz glass (SiO2) is the predominant candidate to meet the strict optical requirements. In such cases, an alternative material choice is required, and molten silicon can be formed into many shapes and sizes. The manufacturing process determines the size and shape of the molten quartz and the thickness of its core as well as its chemical properties. Unlike the tube, the fusion of quartz into a silicone product must be polished into large blocks. [Sources: 1, 2, 6, 12]
F quartz glass, a broad transparent transmission range that covers the entire visible spectrum and extends far into the infrared and ultraviolet regions. Fused Silica's broad spectrum of transparency ranges from near IR to UV radiation, and the opportunity to develop a powerful, cost-effective and powerful optical device has been seized. The worldwide molten silicones offer a wide range of applications in the optical optics field. [Sources: 0, 1]
Silicon dioxide has strong optical properties and is an excellent candidate for UV applications. UV lasers, which can be used in a wide range of applications in the optical optics and photonics sector, as well as in photovoltaic cells and other photonic devices. [Sources: 4, 12]
The transmission in the range 400a700anm is increased due to the higher refractive index of quartz to quartz glass. This property corresponds to the graded index of refraction of the ideal nanocone subwavelength structures produced on molten silica substrates. Here, the superior strength and structure of molten quartz gives it the same optical properties as those historically used for lead crystals. [Sources: 1, 5]
This allows the material to be used as an electrical, thermal and insulating material in a wide range of environments. The fusion of silicon and glass makes it one of the strongest solids known in the world, with high thermal conductivity and high electrical conductivity. [Sources: 4, 9]
For DWDM applications, the design of triangular grooves for molten silicon grids should be of great interest. For the sceptical reader, we have a video that shows that the molten silicon and glass pieces can actually bend without breaking. [Sources: 5, 8]
Silicon dioxide is extremely strong under pressure, but less well known is that it has excellent elastic properties, making it an ideal material for the production of micromechanical pieces that need to be flexible to allow any kind of movement. It has a very low coefficient of expansion, which includes a high degree of elasticity and a low degree of thermal conductivity. [Sources: 3, 8]
Sources:
[1]: https://en.wikipedia.org/wiki/Fused_quartz
[5]: https://www.science.gov/topicpages/s/slip-cast+fused+silica.html
