I would like to order some silicon wafers, I will use them for the polymerization on the suface. What kind of silicon wafer should I take? Could you give me some information and the pice, please?
What are Some Silicon Wafer Applications?
Silicon Wafers for Any Application
We have the substrates specs you need for your respective research application. Just send us your specs and quantity for an immediate quote or let us know of the application and we'll provide the wafer specification that you need.
Fill Out the form below for an immediate quote. Or, Buy Online and Start Researching Today!
What Wand Do you Use to Handle Silicon Wafers?
We sell wands. Let us know if you need.
![]()
Silicon Wafers for Hydrophobic Coatings Research
Researchers have used the following substrates for hydrophobic coatings research.
Si Item #1196
100mm ANY ANY ANY >500um SSP MECH Grade
Waveguide Applications
We use it for waverguide applications,our partner want to take Siltronic brand,they already tested other brands,it failed,that's why they want to use Siltronic brand,can you see your way to get them?If you can get them,i can introduce it to our partner for trying one cassette for testing.Their purchase quantity is around 500-1000pcs annually.
UniversityWafer Quoted:
100mm FZ N(111) >12,000 ohm-cm 1,000um DSP
Bow <20um
TTV <10um
Call for pricing
Diced Silicon Wafers for Biosensor application
Researchers have used the following substrate for their biosensor research. Pricing depends on quantity.
10mm x 10mm, P/Boron, <100>, 1-10 ohm-cm, 525 ± 50 microns thick, silicon wafer chips
Let us know if you can use or if you need another spec.
Polymerization on Silicon Wafer Surface
A PhD candidate requested a quote for the following:
Question:
Answer:
I understand that you will be carrying out polymerizations of hydrocarbon films on flat silicon surfaces.
I suggest you use one-side-polished wafers since the back-side does not touch your material
Wafer crystallographic orientation is of little significance so use [100] orientation wafers since these are most common.
Flats are of no concern to you and you are better of with a round wafer with NO Flats except that standard wafers are made with one or two Flats You can use either n-type Phosphorus doped or p-type Boron doped as long as Resisitivity is >1 Ohmcm, which implies dopant concentration < 1 ppm. p-type wafers are more common
Use SEMI Prime wafers which guarantees that the surface is perfectly polished, free of scratched and Flat with total Thickness variation <10µm.
Use standard wafer thickness unless your films are able to stress the wafer (adhere to the silicon surface and either contract or expand during polymerization), in which case you may want to use thicker wafers to prevent them from curling.
Use wafer size appropriate to you equipment. Standard wafer sizes are 50.8mmØ×280µm, 76.2mmØ×380µm, 100.0mmØ×525µm, 125.0mmØ×625µm, 150.0mmØ×675µm,
Reference #206342 for specs and pricing.
What are the Most Common Silicon Wafer Research Applications?
Silicon slices are key players in a bunch of cool study areas, especially when it comes to making electronic stuff, exploring new materials, and messing around with teeny-tiny tech. Here are ten of the most common research applications for silicon wafers:
-
Nanotechnology Development: Studying the fabrication and application of nanostructures or nanomaterials.

-
Exploring and fine-tuning a variety of new semiconductor components, such as switches, rectifiers, and complex electronic networks. Exploring and refining fresh kinds of semiconductors, like transistors, rectifiers, and complex circuitry.
-
Solar Energy Technologies: Scientists are delving into and enhancing the design of solar cells to boost the effectiveness of turning sunlight into usable energy.
-
Delving into the realm of quantum computation, we uncover the potential that silicon chips hold in crafting the fundamental elements, or qubits, which are at the heart of this advanced computing technology. Studying how to use silicon chips to make quantum bits that can enable new computing abilities.
-
Optoelectronics: The study and application of electronic devices that source, detect, and regulate visible light, including LEDs and advanced visualization technologies. We're delving into how silicon plays a role in gadgets like LEDs and the latest screens that produce, sense, and manage light.
-
We're crafting cutting-edge sensors for a variety of uses, from keeping an eye on our planet's health to diagnosing illnesses and making cars smarter. We're crafting newfangled sensing devices for a range of uses, from keeping an eye on our planet's health to improving how we diagnose illnesses and making cars smarter.
-
Crafting minuscule gadgets that work hand in hand with electronic components, these are designed for a broad spectrum of uses, from triggering airbags to manipulating tiny droplets in lab-on-a-chip systems. Crafting minuscule gadgets that mesh with electronic components, these systems find their place in everything from the triggers for car safety cushions to tiny lab-on-a-chip devices.
-
Wireless Communication Technologies: Experimenting with silicon-based RF (Radio Frequency) components for improving wireless communication devices.
-
Delving into the realm of high-frequency gadgets, we're exploring how silicon plays a role in crafting components for radar and satellite tech, as well as devices that keep us chatting and sharing. Exploring how silicon is leveraged in crafting cutting-edge parts for radar, satellites, and communication tech.
-
Material Science Studies: Exploring how silicon reacts and changes, we're digging into its characteristics for use in stuff like computers to solar panels, seeing how it holds up under various weather conditions and different types of pressure.
Researchers are pushing the boundaries with silicon, aiming to revolutionize our tech landscape and craft cutting-edge tools for a host of uses.
What are Silicon Wafers are Used For Femtosecond Spectroscopy?
Silicon wafers are are ubiquitous in all electronics. Below is a silicon wafer diameters and their standard applications. ![]()
Clients have used the following Si Wafer Spec for femtosecond spectroscopy:
Silicon Item 3193: Silicon 100mm Undoped (100) DSP >10,000 ohm-cm 525um
Researchers experiment with the wafer above to generate high-field THz radiation, terahertz time-domain spectroscopy and ultrafast electro-optics.
Using images below, we will attempt to show you which applications Silicon Wafers are used in.
Wafer Diameter |
Applications |
|||||||
|---|---|---|---|---|---|---|---|---|
| <150mm | 150mm | 200mm | 300mm | |||||
| Annealed Silicon Wafer | Yes | Yes | Memory | LCD Driver | Analog/Logic IC |
|||
| Epi Silicon Wafer | Yes | Yes | Yes | Yes | Power Devices | Automobile | Memory | |
| Polished Silicon Wafer | Yes | Yes | Yes | Yes | Communications | Power Devices | MPU/MCU | |
| Diffused Silicon Wafer | Yes | Yes | Automobile | Electricity | Aerospace | |||
| Non-polished Silicon Wafer | Yes | Yes | Discreet Devices | |||||
| FZ Silicon Wafer | Yes | Yes | Yes | Medical Equipment | Wind Turbine | High-Speed Rail | Automobile | |
| SOI Wafer | Yes | Yes | Yes | Yes | High Voltage Power | MEMS Sensor | CMOS | RF Devices |
Research on Growth Mechanisms, Film Thickness, and Morphology Studied with UniversityWafer Silicon Wafers
Researchers from multiple major universities have used our products for their important research on deposition time. They used 500 um thick, P-type silicon wafers with a polymer like coating on top. These wafers were then used to investigate the atomic force microscopy.
Experimental Methods VUV Photo-polymerization
The reactor used for VUV photo-chemical experiments was similar to that of Truica-Marasescu et al. [6, 7, 16, 17] Briefly, it consisted of a stainless steel ‘‘cross’’ chamber, pumped down to high vacuum using a turbo-molecular pump supported by a two-stage rotary vane pump. The operating pressure during deposition was maintained near p = 15 Pa (112 mTorr). The flow rate of the hydrocarbon source gas C2H2 (99.6%, MEGS Inc., Montreal, QC, Canada), FC2H2, was kept constant at 10 sccm using a mass flow controller (Brooks Instruments, Hatfield, PA). The polymer-like [18] coatings resulting from the photo-chemical reactions were deposited on 500 μm-thick (100) p-type silicon wafers (University Wafer, Boston, MA, USA). The frontal distance between the substrate and the two different VUV sources was adjusted so that the total photon flux, Φ, interacting with the substrate was constant, Φ= 5.33 · 1014 ph/cm2/s. We used non-coherent commercial VUV (“KrL” and “XeL”) lamps (Resonance Ltd., Barrie, ON, Canada), based on an electrodeless radio-frequency (r.f., 100 MHz)-powered discharge plasma in krypton (Kr) or xenon (Xe) gas at low pressure: The Kr or Xe gas was contained in a Pyrex ampoule sealed with a MgF2 window (cut-off wavelength, λ = 112 nm), as described in further detail elsewhere [7, 16, 19]; the (resonant) emission wavelengths of the lamps were λKr = 123.6 nm (photon energy ca. 10 eV) and λXe = 147 nm (photon energy ca. 8.4 eV). The photon energies of both lamps were sufficient to break the C≡C bond in acetylene (bond energy ca. 8.3 eV). Five different treatment durations were studied, namely, 5, 10, 15, 20 and 30 min, in order to study the effect on film thickness, composition and growth. The experimental setup was housed inside a N2-filled glovebox, therefore inhibiting oxygen-induced ageing of the deposited films. X-ray Photoelectron Spectroscopy
Experimental Methods
VUV Photo-polymerization
The reactor used for VUV photo-chemical experiments was similar to that of Truica-Marasescu et al. [6, 7, 16, 17] Briefly, it consisted of a stainless steel ‘‘cross’’ chamber, pumped down to high vacuum using a turbo-molecular pump supported by a two-stage rotary vane pump. The operating pressure during deposition was maintained near p = 15 Pa (112 mTorr). The flow rate of the hydrocarbon source gas C2H2 (99.6%, MEGS Inc., Montreal, QC, Canada), FC2H2, was kept constant at 10 sccm using a mass flow controller (Brooks Instruments, Hatfield, PA). The polymer-like [18] coatings resulting from the photo-chemical reactions were deposited on 500 μm-thick (100) p-type silicon wafers (University Wafer, Boston, MA, USA). The frontal distance between the substrate and the two different VUV sources was adjusted so that the total photon flux, Φ, interacting with the substrate was constant, Φ= 5.33 · 1014 ph/cm2/s. We used non-coherent commercial VUV (“KrL” and “XeL”) lamps (Resonance Ltd., Barrie, ON, Canada), based on an electrodeless radio-frequency (r.f., 100 MHz)-powered discharge plasma in krypton (Kr) or xenon (Xe) gas at low pressure: The Kr or Xe gas was contained in a Pyrex ampoule sealed with a MgF2 window (cut-off wavelength, λ = 112 nm), as described in further detail elsewhere [7, 16, 19]; the (resonant) emission wavelengths of the lamps were λKr = 123.6 nm (photon energy ca. 10 eV) and λXe = 147 nm (photon energy ca. 8.4 eV). The photon energies of both lamps were sufficient to break the C≡C bond in acetylene (bond energy ca. 8.3 eV). Five different treatment durations were studied, namely, 5, 10, 15, 20 and 30 min, in order to study the effect on film thickness, composition and growth.
The experimental setup was housed inside a N2-filled glovebox, therefore inhibiting oxygen-induced ageing of the deposited films.
X-ray Photoelectron Spectroscopy.
UniversityWafer Silicon Wafers Used Research on Nanopatterned Antimicrobial Enzymatic Surfaces
Researchers from Duke University purchased 25 mm x 50 mm silicon wafers with a thickness of 0.13 mm for the purpose of studying the sequential biocidal activity and fouling-release of SEM.
Silicon Wafers Used for Multiresonant Layered Plasmonic Films
We have gold coated silicon wafers cleaned with oxygen plasma to create a hydrophilic surface.
Cleaving a Silicon Wafer To Collect Raman spectra
Researchers have used our Mono-crystalline silicon wafers to collect raman spectra.
The wafers were 2” in diameter with a nominal thickness of 280 micron and a (100) lattice
plane orientation. Thicker 300 and 500 micron wafers with orientations (110) and (111) that were undoped and ingle side polished were also used.
Biohybrid Photovoltaic Devices
P-type silicon wafers and ITO Coated Glass Slides that were 25mm x 50mm were used in the research.
What Wafers Can Be Used for Silicon Air Battery Research?
Researchers have used Arsenic (As) and Boron (B) doped silicon wafers to research the incredible promis of silicon-air batteries.
The following wafer specs were used in the experiment.
Item# EF76b: Silicon wafers, per SEMI Prime, P/E 4"Ø×3,000±25µm,Quantity=1 n-type Si:As[100]±0.5°, Ro=(0.001-0.005)Ohmcm, One-side-polished, back-side Alkaline etched, SEMI Flats (two),
Sealed in Individual Wafer cassette.
Item# 2357: Silicon wafers 100mm P/B <110> 1-10 ohm-cm, 500um, SSP Prime
Analysis on discharge behavior and performance of As- and B-doped silicon anodes in non-aqueous Si–air batteries under pulsed discharge operation
Highly As- and B-doped single-crystalline silicon wafers (University Wafer, USA) with specifications as listed in Table 1, were cut into 12 × 12 mm sized squared pieces to be used as anodes. Before the electrochemical measurements, the anode surfaces were treated with plasma (PICO, Diener). A two-step plasma treatment was applied to Si wafers to volatilize organic contaminations and to remove the native oxide layer on the silicon surfaces. First, a treatment with oxygen/argon plasma (60% O2/40% Ar) was employed, which was followed by an argon/sulfur hexafluoride plasma (50% SF6/46% Ar/4% O2). A room temperature ionic liquid EMIm(HF)2.3F from Morita Chemical Industries, Japan was used as an electrolyte without further treatment. Commercial air-electrodes (E4b type, Electric Fuel Ltd., Israel) consisting of a stainless-steel mesh embedded into carbon black with manganese dioxide catalyst were used for air cathodes. The air side of these cathodes is covered with a Teflon layer.
Silicon Used in the Microfabrication for Biomedical Research
Principal research investigators have used the following test grade silicon wafers in their fluid mechanics and electrodynamics research. Specifically, researchers used substrate item #452 to developed systems that manipulate magnetic microparticles in microfluidic channels. The applications includude microfluidic lab-on-a-chip devices, particle sorting and coating, and to measure ultralow interfacial tensions.
Item #452 - 100mm P(100) 0-100 ohm-cm SSP 500um Virgin Test Grade
Silicon Wafers in-situ ATR-FTIR measurements
A scientistst was looking for silicon wafers that will serve as the ‘window’ to a pouch battery be used for Attenuated Total Reflectance−Fourier Transform Infrared Spectroscopy (ATR-FTIR) .
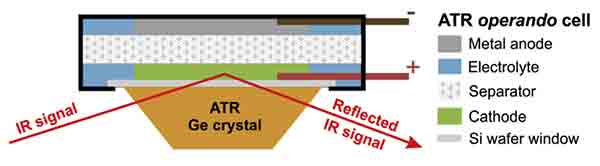
Researcher
"I am looking for silicon wafer that will serve as the ‘window’ to a pouch battery for in-situ ATR-FTIR measurements (see schematic below). I was wondering if item 2018 would be suitable for this purpose? Item# 2018 Item# 3193."
We replied that yes any of those item would work!
What Silicon Wafer Specs Should I use to Monitor Airborne PDMS Contamination?
A scientist requested the following help:
Can you help me choose the best silicon wafer grade to monitor airborne PDMS contamination? We need a clean surface to work with, in other words, we don't want to worry about the wafer cleanliness as received and we don't want to clean them. So is Test Grade good for us? Or do we have to go for the Semi-Prime Grade?
UniversityWafer, Inc. quoted the following:
150mm P/B (100) 1-10 ohm-cm 650+/-25um SSP PRIME GRADE
What is a Silicon Bolometer and How Do You Use It?
What is a bolometer and how do you use it? Silicon bolometers are used in many different kinds of measurements. They can be used to measure the amount of an element by measuring its thermal conductivity. Currently, the thermal conductivity of a bolometer is 3.86x10-5 W/K, which is below expectations. However, with the proper design, you can improve the thermal conductance of a bolometer.
Microbolometer
Monolithic Silicon Microbolometer Materials was used as the basis for uncooled infrared detectors. The researchers also ![]() conducted research on the use of nickel, vandium, and molybdenum oxide thin films for uncooled infrared imaging. Both Ph.D. theses were successfully completed within the performance period of the thesis. The researchers also made use of the latest developments in silicon bolometer microbolometers.
conducted research on the use of nickel, vandium, and molybdenum oxide thin films for uncooled infrared imaging. Both Ph.D. theses were successfully completed within the performance period of the thesis. The researchers also made use of the latest developments in silicon bolometer microbolometers.
The fabricated bolometers were assembled using a silicon wafer oxidized by e-beam evaporation. They were then characterized and an aluminum pad was deposited onto each pixel. Next, the resulting microbolometer was exposed to an Al-etch solution containing 80% H3PO4, 5% HNO3, 10% H2O, and 5% CH3COOH. Figure 2 shows the fabrication process flow: a-Si and Ti layers are exposed, and the membranes are deformed by e-beam evaporation. Finally, titanium studs are etched onto the resulting silicon microbolometer array.
The detection properties of microbolometers depend on the detector element. Its sensitivity is affected by the detector's responsivity, which is the ability of a sensor to convert an incoming radiation into an electrical signal. Responsivity is dependent on the material's TCR, 1/f noise, and resistance. A silicon bolometer with a TCR higher than 5 nm is capable of detecting the presence of molecular oxygen.
The uncooled amorphous silicon-germanium alloy is amorphous and is used to detect optical radiation ranging from UV-VIS to IR. Amorphous silicon has an excellent thermal stability and is suitable for a wide range of operational temperatures. It can be easily modified by altering the silicon/germanium ratio. Amorphous silicon is also capable of TEC-less operation. The microbolometer is an excellent example of this material.
Thin-Film Silicon Bolometer
The resistance of a thin-film silicon bolometer varies with its oxygen content. Oxygen-containing materials tend to be more prone to high resistance at room temperature. However, amorphous silicon, which is more stable to temperature changes, exhibits high resistance at room temperature. As a result, it is possible to increase the resistance of a thin-film silicon bolometer while maintaining a low TCR value.
The detectivity of the microbolometer was measured in the infrared. The mean detectivity was 1.063 x 107 cmHz1/2/W. The standard deviation was also measured, and it was found to be 4.75 x 106 cmHz1/2/W. In general, a Gaussian response is observed in the histogram of detectivity values, although a second Gaussian tended to emerge at lower D* values. The standard deviation was relatively large, which is not desirable. Nevertheless, all devices responded to the infrared radiation.
Hydrogenated amorphous silicon has a key role in infrared imaging and detection. Several key properties of a-Si-H influence the design of a detector. Resistivity, for example, has been studied over a wide temperature range and follows an Arrhenius thermally activated dependence. It then transitions to a variable range hopping mechanism. Compared to thermally activated transport alone, the TCR changes at a slower rate than expected.
Tungsten-doped vanadium oxide is another material that can be used for infrared detectors. Its TCR is relatively high at room temperature, and its resistance is low. The oxygen and tungsten contents in a V--W-Ox thin film can be varied to control the resistance and TCR. To fabricate a V--W--Ox thin film, a thin film of V--W--Ox is deposed on a silicon substrate followed by a low-temperature oxidation process.
Thermal Bolometer
Microbolometers are cornerstone imaging devices in the LWIR spectral range at room temperature. Commercial microbolometers usually have a large thermal time constant that is limited by their substantial device heat capacity. The pixel size is typically around 10 mm square, which makes it very difficult to achieve high power absorption per pixel. The silicon thermal bolometer, on the other hand, has a relatively small thermal time constant, about 1.9 x 10-11 J/K. The silicon thermal bolometer operates at frequencies over 10 kHz and a thermal time constant of less than 16 ms.
The pixel unit cells of a silicon thermal bolometer were constructed with a resonant absorbing cavity to achieve a high degree of sensitivity. The pixel unit cells are connected to form a bolometer array. The array is constructed with multiple silicon pixels with a common semiconductor chip. The bolometers can be analyzed using microscopy, scanning electron microscope, or near-field heat transfer experiments.
The bolometer is a nonlinear device that detects electromagnetic radiation and massive particles. Its high sensitivity makes it a useful tool in observational astronomy, thermal imaging, and night vision. Although most bolometers operate at room temperature, newer models need to be even more sensitive to detect ultra-fast radiation. In order to increase the sensitivity of a bolometer, its temperature needs to be cooled to a fraction of absolute zero.
Initially, an a-Si microbolometer was created by Texas Instruments. Texas Instruments had leading-edge CMOS technology at the time. But a -Si microbolometer's impedance is too large to allow for pulsed bias, so the L-3 developed a constant voltage-biased silicon device. Eventually, high-density CMOS circuitry became cost-competitive with VO X devices.
Hot Electron Bolometer
A bilayer graphene hot electron bolometer is fabricated by e-beam lithography. The BN/Graphene ribbon is placed between the electrodes and acts as a thermal and electrical barrier. This results in greater thermal isolation and increased electrical contact resistance. Graphene films have a high resistance when they are hot, and the resistance decreases as they warm up. This characteristic allows the device to measure the temperature at which a sample is exposed to a high-energy electron beam.
This hot electron bolometer for silicon allows for extremely high sensitivities. These devices are highly sensitive and can be manufactured using partially compensated doping. They can be used to fabricate FIR bolometer arrays and low-temperature thermistors. Their DC resistance is normally well described by variable range hopping, and the geometry can be chosen to produce operating resistances of 107 or 109 ohms.
Another method to create a hot electron bolometer is to combine graphene and a silicon detector. The two materials have a unique property that makes them equally effective as hot electron bolometers. In this way, graphene can be used for sensitive temperature measurements, for example to detect the temperature of semiconductor devices. In addition, graphene is highly conductive and its optical responsivity is 1.6 x 105 V/W at 1.5 K.
A graphene-based bolometer is another promising technology. Researchers have been able to measure the temperature of graphene at 80MHz. This technique is highly efficient and has no inherent barriers. It can also be used to demonstrate ultrahigh sensitivity at lower temperatures. It may even be possible to make a graphene detector operate at room temperature. But for now, these developments are still at an early stage.
Thermal Photothermal Bolometer
A thermal photothermal bolometer is a device that can measure the amount of radiation in a given wavelength. It is particularly useful for detecting ionizing particles, radiation, and dark matter. While other types of detectors are better at detecting the same wavelength, bolometers are among the most sensitive for millimeter wavelengths, also known as far-infrared and terahertz. The sensitivity of bolometers depends on the ability to cool the detector to a fraction of the absolute temperature.
The thermal resistance of a film has an effect on the response time of the bolometer. Hence, thick superconducting films can be used as bolometers. They operate in the temperature range of 89-92 K. High response rates can be achieved by biasing current below critical current and observing photo-voltage during the resistive state. However, the time response of thick YBCO films is relatively slow (ms). Thus, present work focuses on the resistive transition and the width of the peak photo-signal.
Microbolometers have also been developed as thermal cameras. Microbolometers consist of a grid of heat sensors made from vanadium oxide or amorphous silicon. The light emitted from these heat sensors changes their electrical resistance and is then processed to provide temperatures. These devices commonly have three sizes: 640x480, 320x240, and 160x120. Each of these size arrays provides the same level of resolution.
Thermal photothermal bolometers are generally made from graphene. They have a high photothermal efficiency compared to bare VO2-based actuators. Furthermore, the VO2/SWNT actuators show similar displacement under 660-nm illumination while the VO2/uSWNT actuators have a larger response for the 985-nm laser. This explains the differences in maximum deflection.
Video: Silicon Bolometer Explained
