I have purchased wafers from universitywafers, and its pretty good. Now Im looking for wafers with thermal oxide grown on it. I noticed that on your website, there are options for the thermal oxidized wafers with various thickness and growth methods. Id like to know the quality of the 4-inch wafer with 300nm wet thermal oxidized SiO2. Hows the leakage issue of the thermal oxides?
What is Wet Thermal Oxide's Leakage Issues?
An electrical engineer asked the follwoing question regarding leakage issues:
Question:
Answer:
Wet thermal oxide isn’t really designed for insulation/isolation. It is great for photolithography masking, but is too porous to make a good insulator. At 300nm it may work if the conditions are not too extreme. Our recommendation for isolation oxides or gate oxides is Dry Thermal Oxide, especially Dry Thermal Chlorinated Oxide. 300nm of this should take care of leakage. There is the top of the line oxide, Dry Thermal Chlorinated Oxide with an anneal in forming gas, but this isn’t usually needed.
Summary – 300nm of wet oxide may be fine, but it is better to go with 300nm of dry oxide and maybe one of the upgrades.
We don’t have any data on leakage or breakdown voltage. This depends too much on the customer’s processing and on their test method.
Get Your Quote FAST! Or, Buy Online and Start Researching today!
Wet or Dry Thermal Oxide for Gate Dielectric?
A Graduate Research Assistant requested the following quote:
I see that you have p-type silicon wafers that have 300nm of thermal oxide. I have a few questions about that. First I want to make sure that the thermal oxide is in fact SiO2. Is this correct? Also, does this type of wafer come in square shapes? I was hoping to print ZnO materials on this type of wafer in a square pattern. It would be much easier for me to work on a 10 mm by 10 mm wafer rather than a round one. Should I use Wet or Dry Thermal Oxide?
UniversityWafer, Answered:
Definitely Dry Oxide. We have a process for gate dielectrics. It is used for other applications, but gate oxide is the primary purpose. It starts with Dry Chlorinated Oxide and adds a post-oxidation anneal in forming gas.
Dry oxide grows much slower than Wet oxide. This makes it much denser and a better dielectric. The addition of chlorine is to tie up mobile ions in the silicon crystal lattice, especially sodium. It ties them up chemically so that they cannot react with any part of the device or form a leakage pathway. The reason for the anneal in forming gas is to clean up hanging material at the oxide/silicon junction. There are always unfinished silicon chains at the interfaces that can provide a start for a pathway for current. This anneal effectively heals the silicon surface to reduce or eliminate them.
Every part of this process is designed to prevent the formation of a pathway that stray current can use to reach the device. This is ideal for gate oxides. If your client wants to reduce the cost we can do it without the anneal and it will still be a large improvement over wet thermal oxide.
One thing to note is that we cannot process square substrates. Very little semiconductor fabrication equipment can accommodate square substrates. I think you will find this to be true of other foundries.
So we would have to use regular diameters and then dice.
Reference #233769 for specs and pricing.
What Substrates are used for Wet Thermal Oxide?
Wet thermal oxide is a process used in semiconductor fabrication to create a layer of silicon dioxide (SiO2) on a silicon substrate. The process involves exposing the silicon substrate to a wet oxidizing ambient, usually in the form of steam or a mixture of steam and oxygen gas.
The most common substrate used for wet thermal oxide is a single crystal silicon wafer. The wafer is typically doped with either p-type or n-type impurities to create the desired electronic properties for the final device.
The thickness of the oxide layer can be controlled by adjusting the temperature and duration of the oxidizing process. The resulting oxide layer can range in thickness from a few nanometers to several microns, depending on the application.
Other substrates, such as silicon on insulator (SOI) wafers, can also be used for wet thermal oxide. SOI wafers have a thin layer of silicon dioxide sandwiched between two layers of silicon, allowing for the creation of a buried oxide layer during the oxidation process.
Related Thermal Oxide and Silicon Wafer Products
- Thermal Oxide Silicon Wafers
- Silicon Wafers
- Silicon-on-Insulator (SOI) Wafers
- Epitaxial Silicon Wafers
- Wet vs Dry Thermal Oxidation
UniversityWafer, Inc. offers wet thermal oxide coated silicon wafers with a variety of benefits. Our wafers are manufactured using high-quality materials and processes to provide customers with consistent and reliable performance. The benefits of our wet thermal oxide coated silicon wafers include:
-
High-quality oxide layer: Our wet thermal oxide process creates a high-quality silicon dioxide layer on the silicon substrate, providing excellent electrical insulation properties and surface passivation.
-
Customizable thickness: We offer wet thermal oxide coated silicon wafers with a wide range of oxide layer thicknesses to meet your specific application needs.
-
Excellent uniformity: Our wafers have a high degree of uniformity across the wafer surface, ensuring consistent performance across all devices.
-
Wide range of sizes and specifications: We offer wet thermal oxide coated silicon wafers in a variety of sizes and specifications, allowing customers to choose the best option for their application.
-
Competitive pricing: Our wet thermal oxide coated silicon wafers are competitively priced, providing customers with high-quality products at an affordable price point.
What is Wet Thermal Oxide?
Wet thermal oxide is a type of oxide film that is formed on the surface of a silicon wafer when it is exposed to a high-temperature steam atmosphere. The wet thermal oxidation process involves the reaction of silicon with oxygen in the presence of water vapor to produce a silicon dioxide (SiO2) film on the wafer surface. This process is commonly used in the manufacture of semiconductor devices such as microprocessors, memory chips, and solar cells. The wet thermal oxide film has excellent electrical insulation properties and can be used as a protective layer for the underlying silicon substrate.
The wet thermal oxide on silicon wafer (WTO) process is a chemical process which produces a thin oxide layer on a silicon wafer. This oxide layer is a significant factor in the electrical properties of a wafer. This layer is often used in the fabrication of electronic devices, such as microprocessors and transistors. The wet process is considered to be an efficient and inexpensive way to produce high-quality semiconductors.
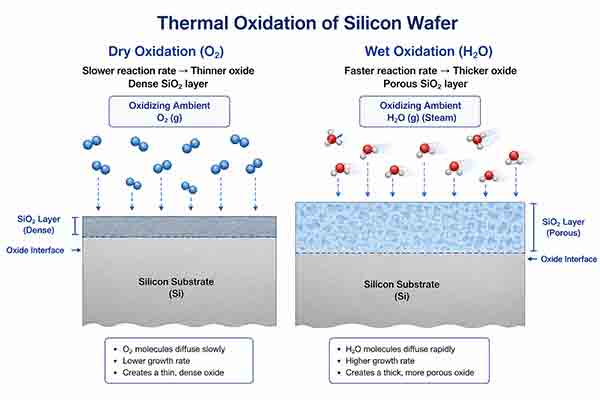
Dry oxidation vs wet oxidation
There are two types of thermal oxidation of silicon, wet and dry. Each type of oxidation has its own advantages. Wet oxidation has a higher growth rate and can produce thicker oxide layers, whereas dry oxidation is more controlled.
The thermal oxidation of silicon occurs when a silicon wafer is exposed to a high temperature. This allows the oxygen atoms to diffuse into the silicon substrate and react with the silicon atoms to form SiO2.
Wet oxidation is often used for thick oxide layers. This type of oxidation is fast and produces a porous oxide film. Wet oxidation also produces hydrogen gas, which is a byproduct of the oxidation process. This is because the water molecule is smaller than the oxygen molecule, allowing it to diffuse through the silicon dioxide layer faster.
Wet oxidation is usually performed at a high pressure. The pressure can be as high as 20 atmospheres. The oxidation rate depends on the crystal orientation of the silicon wafer. For example, the oxidation rate is higher on a (111) oriented surface than on a (100) oriented one.
When wet oxidation is applied to a silicon wafer, the oxidizing gas is supplied to the wafer through a torch chamber. The gases are then fed into a furnace tube through a source end. The heating coils in the furnace tube are generally multiple. The reaction chamber is also heated to an elevated temperature. This serves as an oxidation accelerator. It is possible to vary the hydrostatic pressure in the reaction chamber.
A number of factors affect the quality of the thermally oxidized silicon. For instance, defects on the silicon surface can affect the quality of the oxide. It is important to use a non-reclaim Silicon start material to minimize the risk of contamination.
The temperature of the oxidant is also a factor. Higher temperatures allow more oxygen to diffuse into the silicon substrate. This increases the oxidation rate, which results in thicker oxide layers per unit time. However, high temperatures are also undesirable for multi-step oxidation processes, due to the risk of stacking faults.
Chemical reactions that take place during oxidation
Thermal oxidation is a process that transforms bare silicon wafers into thin and dense oxide layers. This chemical reaction is necessary for the fabrication of silicon wafers. It provides passivation, chemical protection, and electrical isolation between layers.
The process is typically done in a furnace. Typical oxidation furnaces are hot wall resistively heated quartz tubes. The process is usually carried out at temperatures between 900 and 1200 degrees Celsius.
The chemical reaction occurs when oxygen and silicon interact at the silicon/silicon dioxide interface. In this case, the bond strength between the silicon and oxygen is covalent and strong. This makes it possible to use the resulting oxide to gain control over the band gap of the semiconductor. In addition, this chemistry provides a barrier against corrosion.
The rate of oxidation depends on the concentration of oxidants and the velocity of the diffusion of the oxidant through the oxide layer. This reaction is controlled by the processes that take place in the silicon surface.
The growth of the oxides on the silicon wafer surface is dependent on the pressure, temperature, and orientation of the substrate. The uniformity of the layer is also determined by these factors. The thickness of the oxide film is limited by its exponential growth. The density of the layer varies according to the oxidation and annealing conditions.
The growth of the oxides on a silicon wafer is a very tedious and time-consuming task. It is important to produce uniform and pure oxides. A number of different methods for growing oxides can be used. The wet oxidation method is generally preferred because it produces denser films. However, the dry oxidation method is also used in some applications.
The wet oxidation technique uses clean steam. In some cases, other gases are used to remove impurities. These gases include nitrogen and hydrogen. In the laboratory, HCl is not used because of the risk of hazardous gas exhaust.
A new design of water vapor delivery systems is being introduced. This eliminates the need for pyrolytic torches, water bubblers, and other devices. The benefits of using this design include lower cost and improved uniformity.
Electrical properties of the resulting oxide layer
Thermal oxidation is the process of adding oxygen atoms to silicon to create silicon dioxide. It is an important technique in microelectronics, since it produces an oxide layer which has low defects. In addition, it provides isolation between neighboring devices. This technique is also used to reduce reverse current.
There are two ways to oxidize silicon. The first is dry oxidation. This involves using an external torch. The second is wet oxidation, in which hydrogen and oxygen gases are used. Wet oxidation is preferred when producing thick oxides.
The difference between wet and dry oxidation is that wet oxidation produces porous films. This means that the thickness of the oxide film can be determined at a particular time. In addition, wet oxidation is faster.
The growth rate is proportional to the thickness of the oxide layer. The thickness of the layer depends on the amount of oxygen atoms that are present in the silicon wafer. It is also determined by the orientation of the silicon crystal. If the crystal is oriented at the top, the oxidation rate is higher than if the crystal is oriented at the bottom.
The oxidation rate is governed by the valence electrons that are shared between silicon and oxygen. The resulting oxide layer has the potential to act as a gate dielectric for MOS devices. In addition, it can help mask impurities. It can also serve as a protective shield for silicon during implantation.
In addition to serving as a protective shield, oxides can be used to help passivate the surface of silicon. However, these properties are only effective if the silicon is partially converted to the oxide.
One reason for this is that the oxidation speed is influenced by the lateral diffusion of oxidant molecules through the oxide layer. It is important to ensure that the oxidation temperature is high enough to produce a viscous flow. If the temperature is too high, the oxidant molecules may diffuse across the oxide layer and into areas not intended for oxidation. This can result in the formation of warpage and bows in the wafer.
Process for the preparation of curved silicon nanowires
Silicon nanowires are extremely beneficial for applications in transistors and sensors. They have excellent electrical and mechanical properties. They can be made in a variety of ways. Several methods have been developed for preparing silicon nanowires.
Silicon nanowires can be produced by removing bulk silicon or through catalyzed growth. The process is simple and relatively low-cost. It enables the manufacture of large-area silicon nanowire structures at normal temperatures. It can be applied to virtually any one-dimensional inorganic nanomaterial.
The new method of preparing silicon nanowires on silicon wafers is inexpensive, easy to use, and does not require a complicated template. It can be used in a variety of applications, including electronics of extremely sensitive biosensors. It also enables the manufacture of novel network materials.
To produce silicon nanowires, the silicon substrate is dipped in an aerated HF solution, and then dried by N2 blow. Silicon particles are precipitated out of the metal silicon droplets, and a thin layer of silicon is formed. This layer increases the motility of silicon nanowire holes. The thickness of the nanowire can be varied, and the length of the wires can be controlled.
The surface chemistry of the Si nanowires determines their biosensor performance. SiNWs prepared by 20% H2O2 etching solution exhibit the best activity in decomposing organic pollutants. The growth rate depends on the catalyst particle size.
Chemical vapour deposition is another common technique for enhancing the growth of silicon nanowires. It is performed using catalyst aerosols or catalyst nanoparticles in a gas stream. The catalyst nanoparticles inject holes into the substrate, which forms macroscopic networks. The density and diameter of the nanowires are determined by the concentration of the metal salts.
Various groups have reported the use of high-control patterned metal films for assembling silicon structures. This approach, which combines lithography and self-assembly, can be used to fabricate honeycomb arrays, vertically aligned silicon structures, and tilted silicon structures. It can also be combined with a self-assembly step to fabricate silicon gratings.
The oxidation process of silicon nanowires is self-limiting, which means that the amount of oxidant used can be accurately controlled. This makes it possible to manufacture Si nanowires with high aspect ratios and low-diameters.
